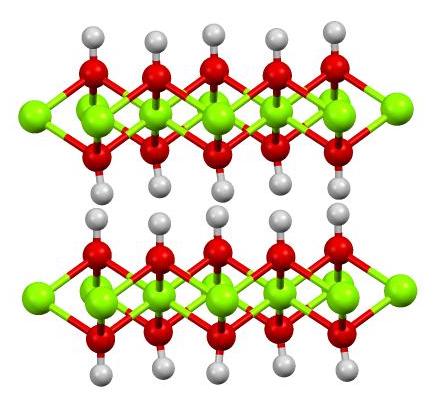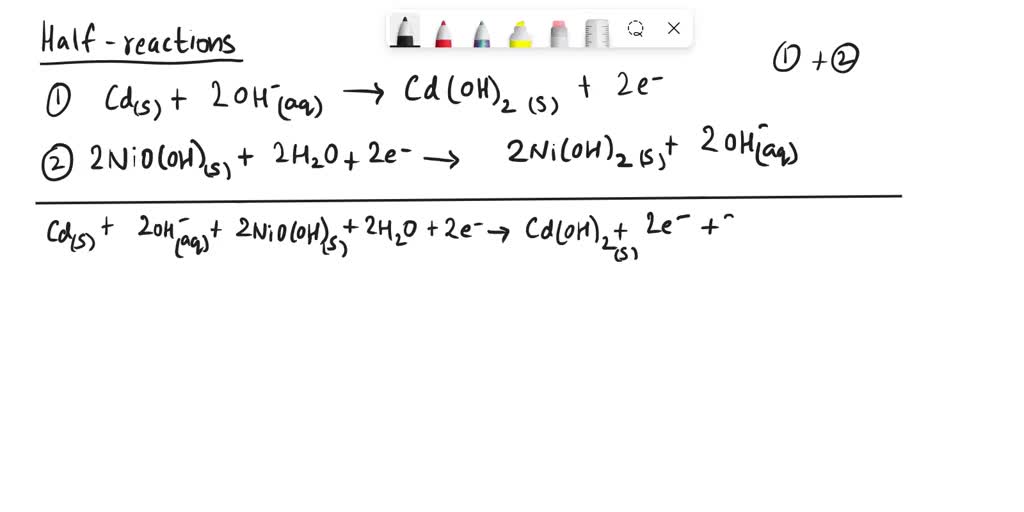
SOLVED: The half reactions in NiCad (nickel-cadmium) batteries are: Cd(OH)2(s) + 2e- â†' Cd(s) + 2OH- (aq) 2NiO(OH)s + 2H2O + 2e- â†' 2Ni(OH)2(s) + 2OH-(aq) Write the net ionic equation for
Cadmium hydroxide, China Cadmium hydroxide, Cadmium hydroxide exporters, China Cadmium hydroxide Suppliers - zhonghe-chem
The molar solubility of Cd(OH)2 is 1.84 × 10^–5 M in water. The expected solubility of Cd(OH)2 in a buffer solution of pH = 12 is : - Sarthaks eConnect | Largest Online Education Community
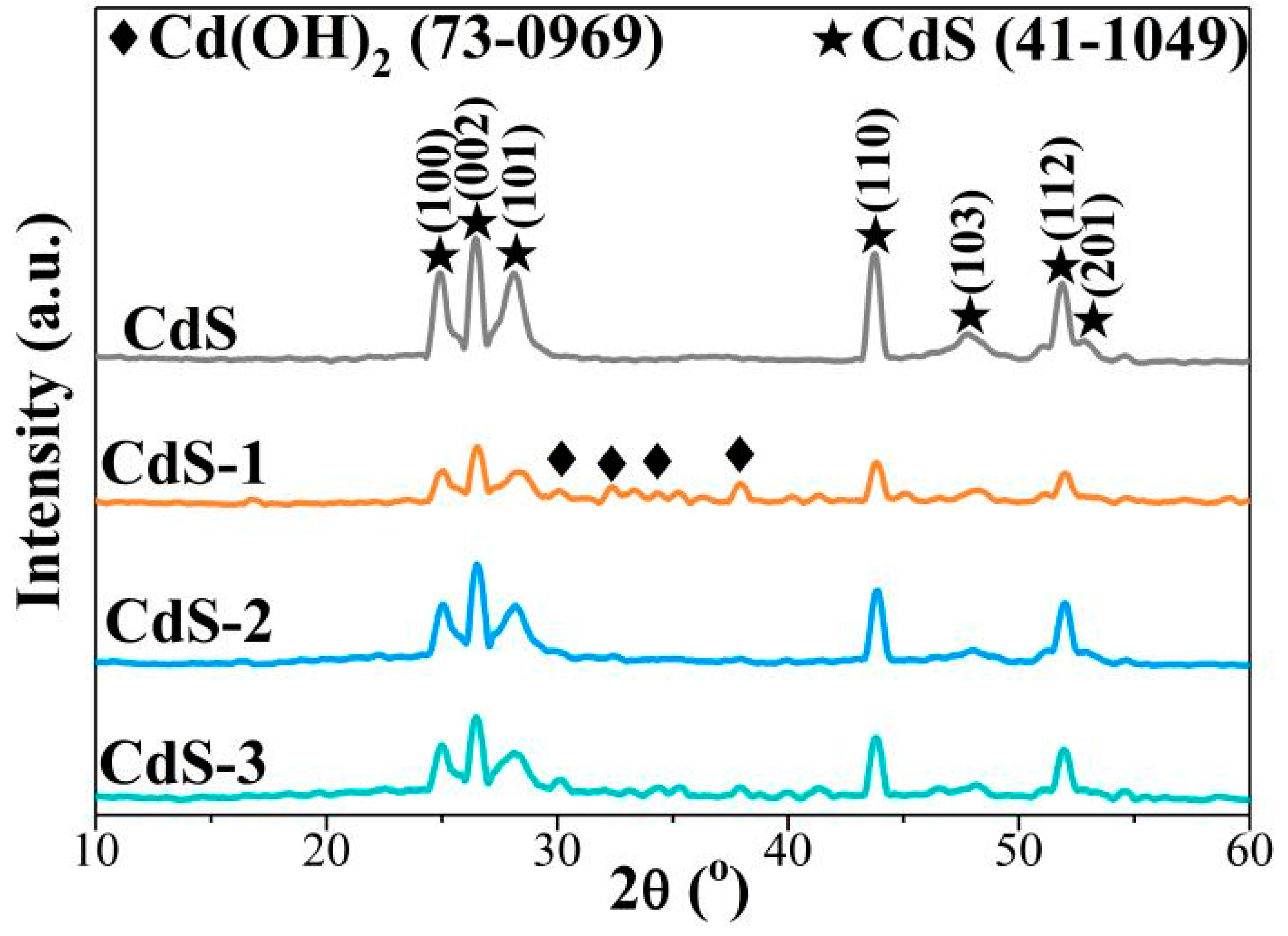
Nanomaterials | Free Full-Text | In Situ Fabrication of CdS/Cd(OH)2 for Effective Visible Light-Driven Photocatalysis
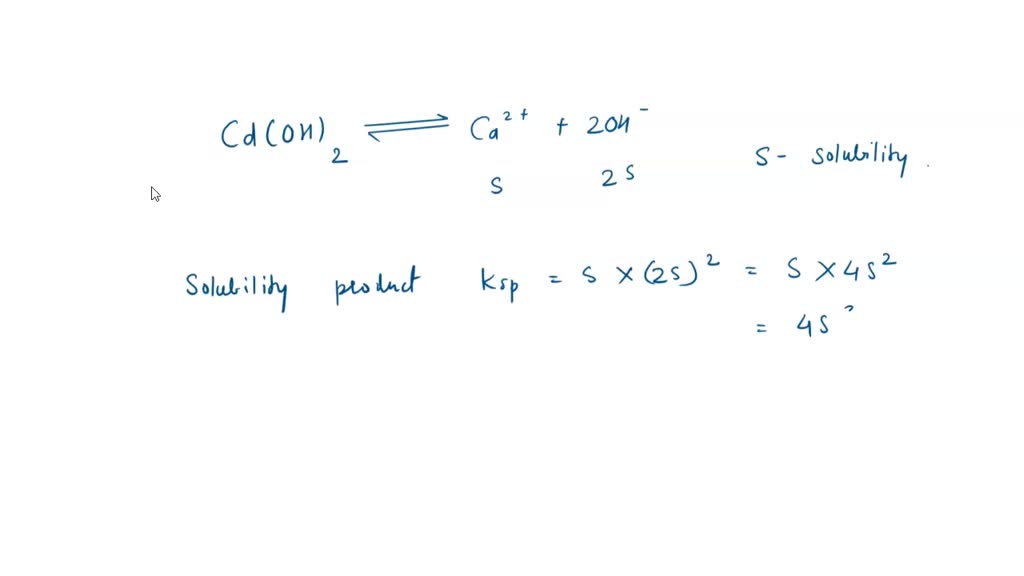
SOLVED: The solubility product constant for Cd(OH)2 is Ksp = 2.5 × 10^(-14) at 25 °C. a. What is the molar solubility of Cd(OH)2? b. What is the mass solubility of Cd(OH)2

SOLVED:Cd(OH)2 is an insoluble compound. It dissolves in excess NaOH in solution. Write a balanced ionic equation for this reaction. What type of reaction is this?

Remarkable positive effect of Cd(OH)2 on CdS semiconductor for visible-light photocatalytic H2 production - ScienceDirect
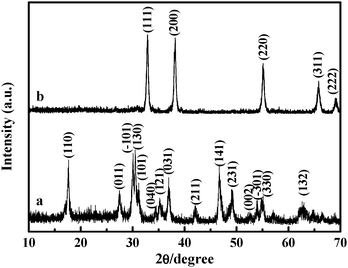
One-step fabrication of Cd(OH)2 nanorings via a solution phase synthesis - Chemical Communications (RSC Publishing) DOI:10.1039/C0CC00665C

Solubility of `Cd(OH)_(2)` in pure water is `1.84xx10^(-5)\"mole\"//L` Calculate its solubility in - YouTube